
BOA™ Biomedical’s GARNET™ therapeutic product begins First in Human clinical trial
BOA Biomedical’s GARNET™ therapeutic product begins FIH clinical trial
Addressing the unmet medical needs of patients with a serious or life-threatening disease or condition is important to BOA Biomedical. BOA Biomedical does maintain an Expanded Access program for circumstances where a patient with a life-threatening illness, such as COVID-19, requires treatment and the treating physician believes that the GARNET technology may be of benefit.


Recognize active infection and assess infection severity and sepsis within minutes in any setting, including the home.
Accurately identify infection-causing pathogens, within one hour, without the need for blood cultures.
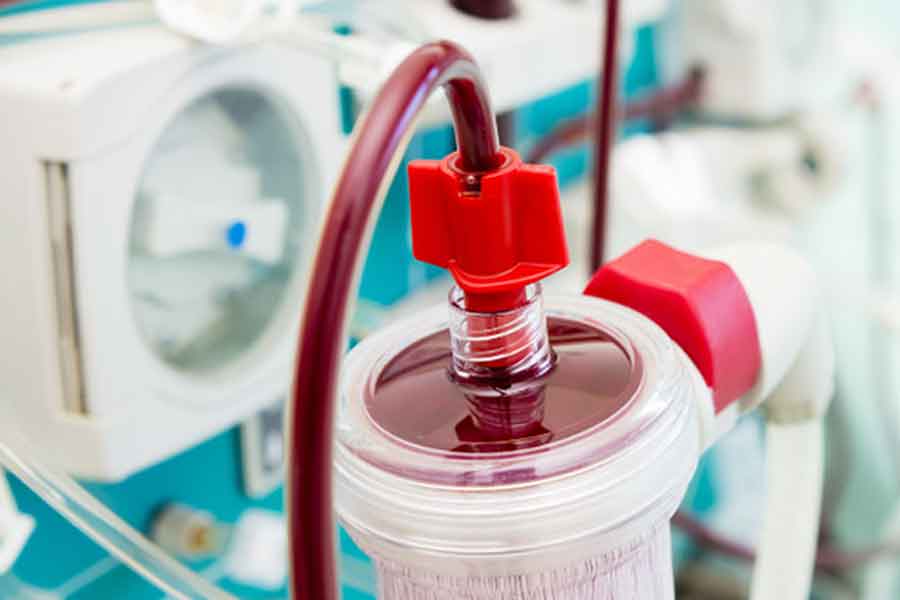
Filter the toxic by-products of pathogens directly from a patient’s blood using an extracorporeal therapeutic filter.

BOA, DOD and their research agency, DARPA, focus research on diagnosing and treating sepsis. BOA licensed the technology from Harvard University in 2018 and was awarded emergency grants from the Department of Defense in 2020.

BOA Biomedical’s GARNET™ therapeutic product begins FIH clinical trial

The study, expected to begin later this year, will take place at Walter Reed National Military Medical Center and at nonmilitary hospitals.

Harvard’s Wyss Institute and VC firm Miraki Innovation have created a joint venture BOA Biomedical, which is developing a device that could diagnose and treat
A Cambridge biomedical is developing a rapid coronavirus test that it hopes to have ready in a few months.